Year 7 Lab Equipment Quiz
True / False
True / False
Select all that apply
True / False
True / False
Select all that apply
Select all that apply
Select all that apply
Put in order
Frequent Equipment Selection Errors in Lab Heating Questions
Frequent Equipment Selection Errors in Lab Heating Questions
Questions about bringing one liter of water to 85°C test your understanding of how specific pieces of scientific equipment are used together. Learners often know each item by name but still choose the wrong combination. The mistakes below explain why some options are incorrect and how to think through similar problems.
Using a balance instead of a thermometer
Many students pick a balance when they see one liter of water mentioned. A balance measures mass, not temperature. The task is to reach 85°C, so a thermometer is essential. Use a balance only if the question asks for mass in grams, not for temperature control.
Heating water directly in a graduated cylinder
Graduated cylinders are for accurate volume measurement, not for heating. The glass is tall and narrow, which increases risk of tipping and uneven heating. Correct reasoning uses the graduated cylinder to measure one liter, then transfers the water to a beaker for heating on a hot plate.
Choosing a test tube for large volumes
Test tubes are for small samples, usually a few milliliters to tens of milliliters. One liter of water cannot safely fit in a test tube, and even partial filling would heat very unevenly. For one liter, a large beaker is the appropriate container.
Ignoring the need for controlled, uniform heating
A Bunsen burner gives intense, localized heat. This is poor control for a target temperature like 85°C in a large volume. A hot plate provides more even heating and easier adjustment. When you see a precise target temperature for a big volume, prefer a beaker plus hot plate, monitored with a thermometer.
Lab Equipment Quick Reference for Heating One Liter of Water
Lab Equipment Quick Reference for Heating One Liter of Water
Note: You can print or save this sheet as a PDF for quick reference during lab preparation or quiz review.
Key pieces of equipment and their main functions
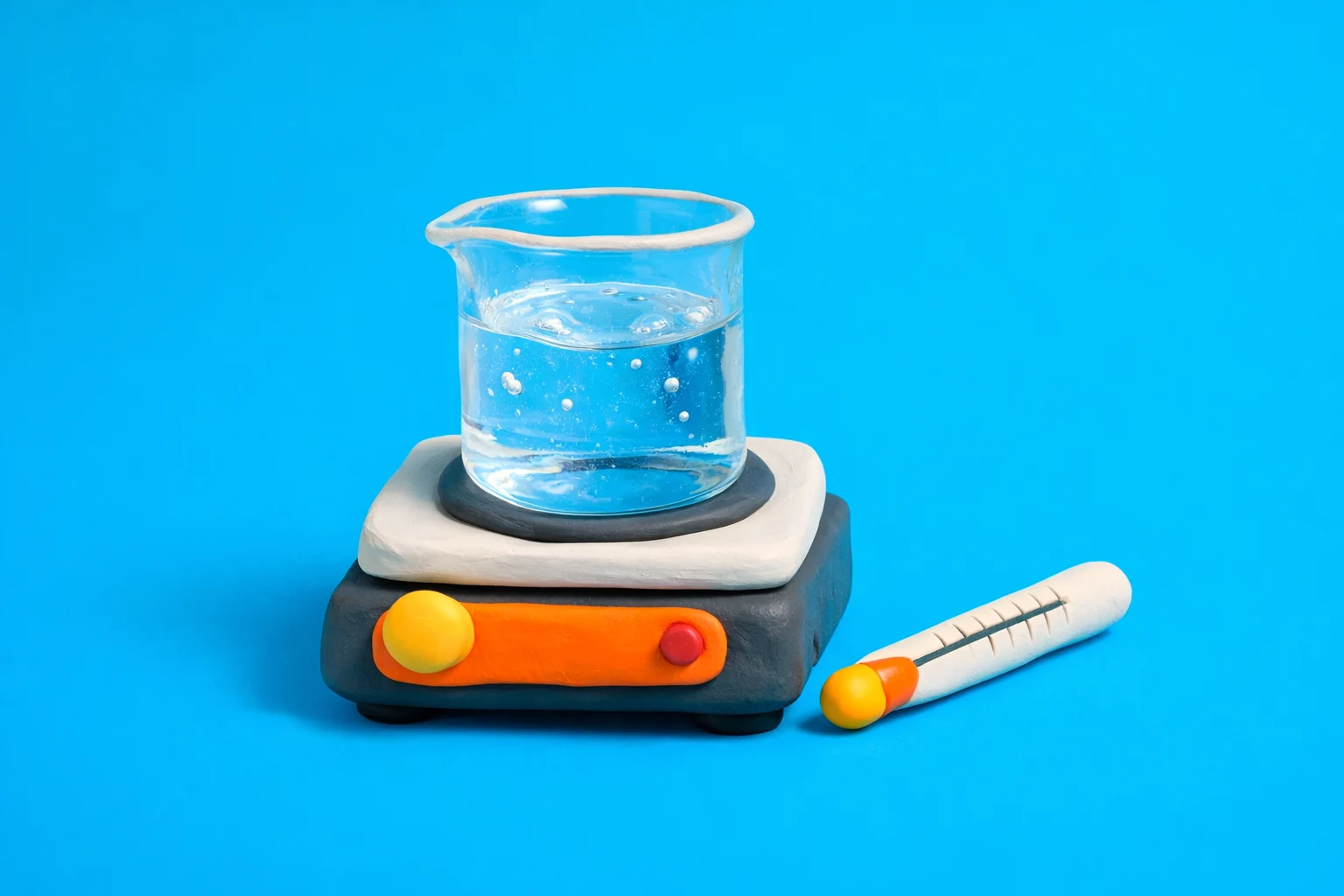
- Graduated cylinder: Measures liquid volume accurately. Use it to measure one liter of water before transfer to a beaker.
- Beaker: Holds liquids for heating, mixing, and rough volume estimates. Wide shape allows even heating on a hot plate.
- Thermometer: Measures temperature. Needed to know when the water reaches 85°C instead of boiling or staying too cool.
- Hot plate: Provides adjustable, even heating under a beaker or flask. Safer and more controlled than an open flame for large volumes.
- Balance or electronic balance: Measures mass, for example grams of a solid. Not used to measure temperature.
- Test tube: Holds very small volumes for reactions or heating small samples. Not suitable for one liter of water.
- Bunsen burner: Produces a flame for high temperature heating of small items. Gives less control for reaching a specific moderate temperature in a large volume.
Standard procedure to heat one liter of water to 85°C
- Use a graduated cylinder to measure exactly one liter of water.
- Pour the water into an appropriate beaker that can safely hold more than one liter.
- Place the beaker on a hot plate.
- Insert a thermometer so the bulb is fully in the water, not touching glass or the hot plate surface.
- Turn on the hot plate to a moderate setting. Stir gently if allowed for more even heating.
- Watch the thermometer closely. Turn off the hot plate or remove the beaker when the temperature reaches 85°C.
Quick decision rules for quiz questions
- If the task specifies a target temperature, you must include a thermometer.
- If the volume is large (hundreds of milliliters or more), choose a beaker plus hot plate, not a test tube or Bunsen burner.
- Use a graduated cylinder for accurate volume measurement, then heat in a beaker.
- Select a balance only when the question mentions mass or grams, not when it asks for temperature or heating to a set degree.
Step-by-Step Reasoning for Lab Equipment Combination Questions
Step-by-Step Reasoning for Lab Equipment Combination Questions
Example 1: One liter of water to 85°C
Question idea: Many types of scientific equipment are used to perform different functions in the science lab. Which combination is best to bring one liter of water to 85°C?
Step 1: Identify tasks. You must measure volume, hold the water while heating, supply heat, and monitor temperature.
Step 2: Match tasks to tools.
- Accurate volume measurement of one liter requires a graduated cylinder.
- Safe heating of a large volume requires a beaker.
- Controlled heating is provided by a hot plate.
- Monitoring 85°C requires a thermometer.
Step 3: Eliminate distractors. A balance or electronic balance measures mass, which is not requested. A test tube is too small for one liter. A Bunsen burner makes it harder to stop precisely at 85°C. The only fully suitable set is graduated cylinder, beaker, thermometer, and hot plate.
Example 2: Heating 250 mL of solution to 60°C
Step 1: Break down the task. Measure 250 mL, heat safely, and reach 60°C accurately.
Step 2: Choose equipment. Use a graduated cylinder to measure 250 mL. Transfer to a beaker. Place the beaker on a hot plate and use a thermometer to monitor temperature.
Step 3: Reject wrong options. Any combination that replaces the thermometer with a balance cannot confirm 60°C. A test tube and Bunsen burner combination is better suited for small samples and high temperatures, not for controlled heating of 250 mL to a moderate temperature.
By repeating this task matching method, you can analyze any equipment combination question in the quiz.
Lab Heating Equipment Selection Quiz FAQ
Lab Heating Equipment Selection Quiz FAQ
Why is a thermometer required for questions that specify 85°C?
Reaching 85°C is a temperature control task. Visual signs such as steam or small bubbles only give rough hints about temperature. A thermometer provides a numerical reading so you can stop heating exactly at the required value and avoid overheating beyond the target.
Why is a beaker preferred over a test tube for one liter of water?
A beaker can hold large volumes and has a wide base for stability on a hot plate. Heat spreads more evenly across the bottom surface. A test tube is narrow and intended for small samples. It cannot safely hold one liter and would heat very unevenly if you tried.
What role does the graduated cylinder play if the beaker already has markings?
Graduated cylinders provide more precise volume measurements than most beakers. Beaker markings are approximate and often used only for quick estimates. For a question that stresses one liter of water, the best practice is to measure with a graduated cylinder, then move the exact volume into the beaker for heating.
Why is a hot plate usually safer than a Bunsen burner for this type of task?
A hot plate gives an even, adjustable heat source and does not involve an open flame. This reduces risk of burns and fire when using large volumes of liquid. It also helps you change the heating rate more gradually to approach 85°C without overshooting.
When would a balance or electronic balance be more appropriate than a thermometer?
A balance is used when the question asks for the mass of a substance, such as grams of a solid reactant. If you only need to reach a specific temperature, a balance does not help. For temperature targets, you always prioritize using a thermometer over a balance.





